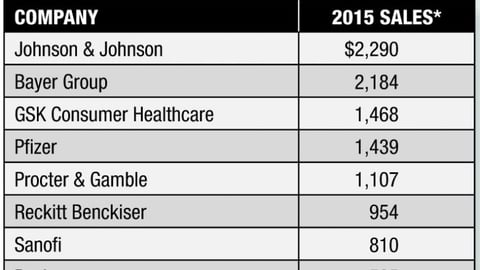
-
Apple disrupts diabetes management with subscription service
NEW YORK — One Drop on Tuesday announced that it has received U.S. Food and Drug Administration 510(k) clearance and Conformité Européene (CE) mark approval for One Drop | Chrome, a blood glucose monitoring system that marries modern design with advanced medical and mobile technology.