-
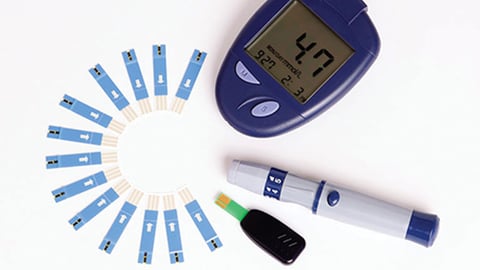
Trend toward self-management fuels growth of diabetes device sales
With the increasing trend of self-management and the rising awareness around the diabetes disease state within developing countries, blood glucose meters can be utilized to aid in adjustment of therapeutic regimen in response to blood glucose values and to help patients adjust their dietary intake, physical activity and insulin doses to improve glycaemic control on a regular basis.
This and several other factors are covered in the latest Research and Marekts report focused on diabetes care devices.
-
Kroger, APhA Foundation to help CDC scale National Diabetes Prevention Program
The Centers for Disease Control and Prevention will be working with the American Pharmacists Association to build infrastructure to expand a program from the National Diabetes Prevention Program within community pharmacies.